Updated on
Summary The outbreak has alarmed US and focused attention on regulations of pharmaceutical companies.
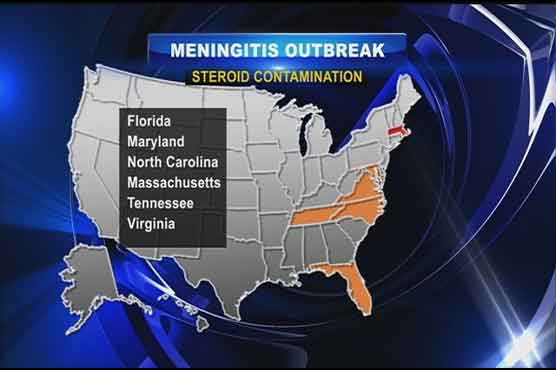
US health officials on Sunday reported an additional 27 cases in a fungal meningitis outbreak linked to steroid injections that has killed seven people and now infected 91 in nine states.The US Centers for Disease Control and Prevention reported the new total of 91 cases in an update on its website, up from 64 on Saturday. Most of the new cases were reported in Michigan, where the total increased to 20 from eight. Virginias total increased to 18 from 11.The widening outbreak has alarmed U.S. health officials and focused attention on regulations of pharmaceutical compounding companies like the one that produced the drugs, the New England Compounding Center Inc in Framingham, Massachusetts.The company shipped 17,676 vials of the steroid methylprednisolone acetate to 76 facilities in 23 states from July through September, the Massachusetts Health Department said. The steroid is used as a painkiller, usually for the back, and could have been injected in thousands of patients, authorities have said.Meningitis is an infection of the membranes covering the brain and spinal cord, and affected patients started showing a variety of symptoms from one to four weeks after their injections.The company, which was previously the subject of complaints, has suspended its operations while an investigation proceeds and earlier recalled the three lots of the drug. It expanded its recall on Saturday to all products compounded and distributed at its Framingham facility.According to an announcement on its website, the company issued the broader recall out of an abundance of caution because of the potential risk of contamination. A compounding pharmacy takes medications from pharmaceuticals manufacturers and makes them into specific dosages and strengths for use by doctors.Complaints against the company in 2002 and 2003 about the processing of medication resulted in an agreement with government agencies in 2006 to correct deficiencies, the Massachusetts Health Department said.





